RadPlanBio: An Integrated Platform for Translational Cancer Research
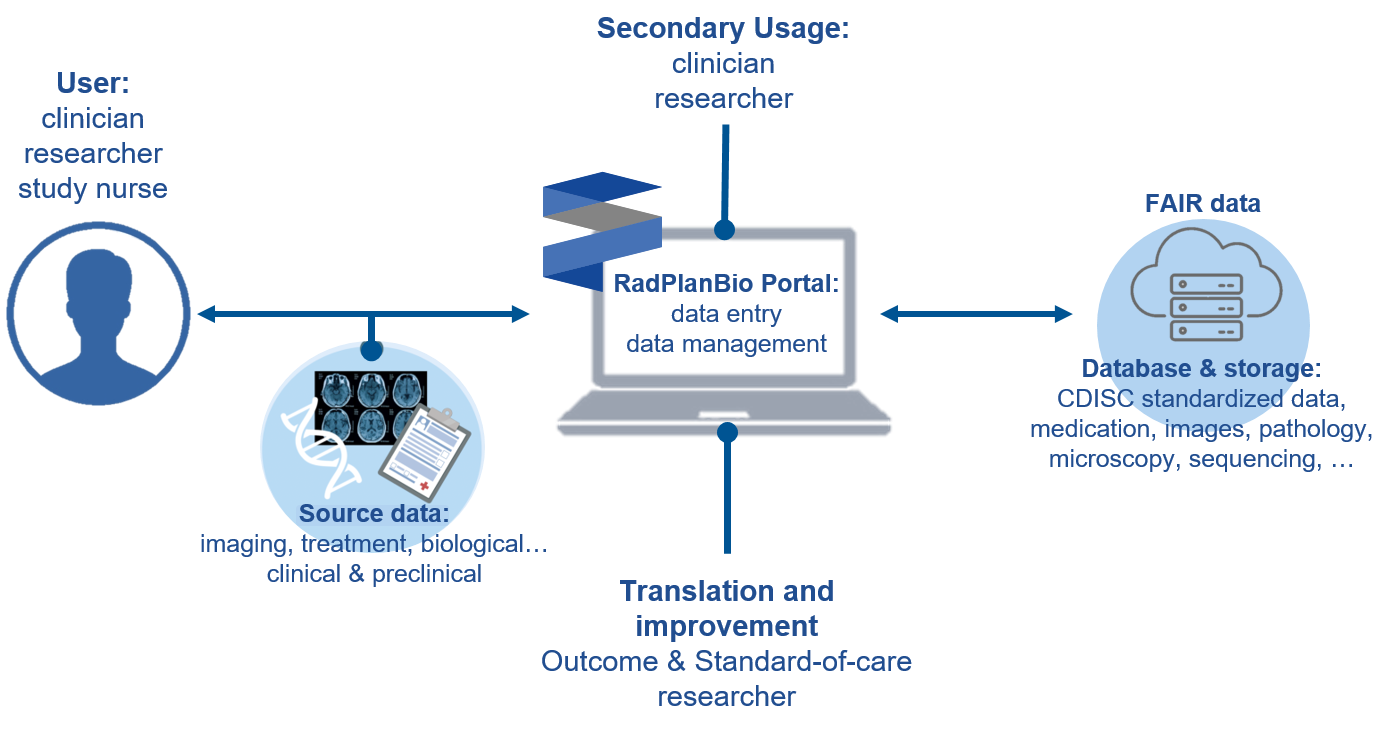
The DKTK RadPlanBio platform offers a comprehensive, web-based data management environment to support both preclinical and clinical cancer research trials within the DKTK and beyond. RadPlanBio accommodates a broad spectrum of translational research needs by integrating diverse data types, such as medical imaging, treatment plans, laboratory assays, genomic data, and clinical outcomes. This makes it well-suited for both clinical investigations and preclinical (including animal) research. By bringing these data together, RadPlanBio enables researchers to more easily relate laboratory findings to patient information and deepen their understanding of disease pathways.
For example, in the interventional DELPHI trial, that evaluates reduced adjuvant therapy for HPV-positive oropharyngeal SCC, RadPlanBio ensures structured data documentation and reliable treatment group assignment. It facilitates dummy runs and treatment plan quality assurance, ensuring protocol adherence across centers. The platform further supports data monitoring and integrates biomarker collection with clinical and radiotherapy data, providing a robust basis for translational analyses.
For preclinical trials RadPlanBio supports both retrospective and prospective study documentation, including transparent treatment group assignment. For example in the NIMO preclinical trial, investigating prognostic biomarkers of response to nimorazole combined with radiochemotherapy in HNSCC xenografts, RadPlanBio enables the determination of tumor control rates and integrates biomarker data such as transcriptomics, methylomics, and histology (hypoxic volume, vessel density, perfusion). By combining these datasets, RadPlanBio provides a robust basis for identifying predictive biomarkers and translating preclinical findings into clinical research.
The platform is continually evolving to incorporate semantic technologies and align with widely accepted standards, including Good Clinical Practice (GCP), CDISC guidelines for data standardization. An electronic case report form (eCRF) system ensures consistent data collection and adherence to clinical standards and data protection protocols. A warehouse stores the collected data in a standardized structure, ensuring compliance to the FAIR principles, helping researchers to efficiently explore and analyse their data.
RadPlanBio is securely hosted at DKTK Dresden and DKFZ Heidelberg in sync. It facilitates multi-site collaborations by enabling data sharing across single- and multi-center clinical and preclinical trials and projects.
If you are interested in studying biomarkers, evaluating therapeutic combinations, or undertaking comprehensive outcomes research, RadPlanBio provides a reliable and trusted infrastructure for your work. The RadPlanBio team welcomes researchers interested in developing their projects within this collaborative environment.
Contact
For all requests, advice or further information please contact: radplanbio@dkfz-heidelberg.de
Coordination Dresden

Prof. Dr. Mechthild Krause
Head of department – partner site Dresden

Dr. Antje Dietrich
Scientific Coordinator Dresden
OncoRay – National Center for Radiation Research in Oncology
Coordination Heidelberg

Registered software:
https://helmholtz.software/software/radplanbio
Further links:
https://www.dkfz.de/radioonkologie-radiobiologie/radplanbio
https://www.oncoray.de/de/netzwerk/deutsches-konsortium-fuer-translationale-krebsforschung-dktk/hnpraedbio-studie-und-radplanbio-plattform
Selected Publications:
Platform
Skripcak T, Belka C, Bosch W, Brink C, Brunner T, Budach V, Büttner D, Debus J, Dekker A, Grau C, Gulliford S, Hurkmans C, Just U, Krause M, Lambin P, Langendijk JA, Lewensohn R, Lühr A, Maingon P, Masucci M, Niyazi M, Poortmans P, Simon M, Schmidberger H, Spezi E, Stuschke M, Valentini V, Verheij M, Whitfield G, Zackrisson B, Zips D, Baumann M. Creating a data exchange strategy for radiotherapy research: towards federated databases and anonymised public datasets. Radiother Oncol. 2014 Dec;113(3):303-9. doi: 10.1016/j.radonc.2014.10.001.
Skripcak T, Just U, Schönfeld I, Troost EGC, Krause M. Storage and databases for big data, chpt. 3, Big Data in Radiation Oncology, 1st Edition CRC press, 2019, https://doi.org/10.1201/9781315207582
Giraldo O, Sabyrrakhim A, Roscher M, Euler-Lange R, Baumann M, Kurth I, Hadiwikarta WW.
Semantic Representation of Preclinical Data in Radiation Oncology. ICBO 2024, Enschede, The Netherlands, July 18-19 2024, CEUR-WS.org, online https://CEUR-WS.org/Vol-3939/short2.pdf
Clinical
Löck S, Linge A, Lohaus F, Ebert N, Gudziol V, Nowak A, Tinhofer I, Kalinauskaite G, Guberina M, Stuschke M, Balermpas P, von der Grün J, Grosu AL, Debus J, Ganswindt U, Belka C, Peeken JC, Combs SE, De-Colle C, Zips D, Baretton GB, Krause M, Baumann M; Biomarker signatures for primary radiochemotherapy of locally advanced HNSCC - Hypothesis generation on a multicentre cohort of the DKTK-ROG. Radiother Oncol. 2022 Apr;169:8-14. doi: 10.1016/j.radonc.2022.02.009.
Tawk B, Rein K, Schwager C, Knoll M, Wirkner U, Hörner-Rieber J, Liermann J, Kurth I, Balermpas P, Rödel C, Linge A, Löck S, Lohaus F, Tinhofer I, Krause M, Stuschke M, Grosu AL, Zips D, Combs SE, Belka C, Stenzinger A, Herold-Mende C, Baumann M, Schirmacher P, Debus J, Abdollahi A. DNA-Methylome-Based Tumor Hypoxia Classifier Identifies HPV-Negative Head and Neck Cancer Patients at Risk for Locoregional Recurrence after Primary Radiochemotherapy. Clin Cancer Res. 2023 Aug 15;29(16):3051-3064. doi: 10.1158/1078-0432.CCR-22-3790.
Martin D, Schreckenbach T, Ziegler P, Filmann N, Kalinauskaite G, Tinhofer I, Budach V, Gani C, Zips D, Schimek-Jasch T, Schäfer H, Grosu AL, Thomas E, Krause M, Dapper H, Combs S, Hoffmann C, Stuschke M, Walter F, Belka C, Kurth I, Hadiwikarta WW, Baumann M, Rödel C, Fokas E. Evaluation of prognostic factors after primary chemoradiotherapy of anal cancer: A multicenter study of the German Cancer Consortium-Radiation Oncology Group (DKTK-ROG).
Radiother Oncol. 2022 Feb;167:233-238. doi: 10.1016/j.radonc.2021.12.050.
Preclinical
José Besso M, Bitto V, Koi L, Wijaya Hadiwikarta W, Conde-Lopez C, Euler-Lange R, Bonrouhi M, Schneider K, Linge A, Krause M, Baumann M, Kurth I. Transcriptomic and epigenetic landscape of nimorazole-enhanced radiochemotherapy in head and neck cancer. Radiother Oncol. 2024 May 30:110348. doi: 10.1016/j.radonc.2024.110348.
Bitto V, Hönscheid P, Besso MJ, Sperling C, Kurth I, Baumann M, Brors B. Enhancing mass spectrometry imaging accessibility using convolutional autoencoders for deriving hypoxia-associated peptides from tumors. NPJ Syst Biol Appl. 2024 May 27;10(1):57. doi: 10.1038/s41540-024-00385-x.
Koi L, Bitto V, Weise C, Möbius L, Linge A, Löck S, Yaromina A, Besso MJ, Valentini C, Pfeifer M, Overgaard J, Zips D, Kurth I, Krause M, Baumann M. Prognostic biomarkers for the response to the radiosensitizer nimorazole combined with RCTx: a pre-clinical trial in HNSCC xenografts. J Transl Med. 2023 Aug 26;21(1):576. doi: 10.1186/s12967-023-04439-2. PMID: 37633930